

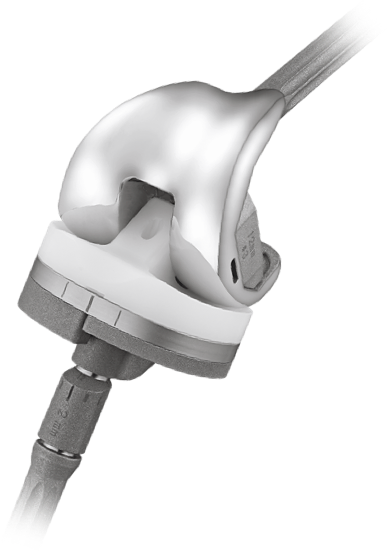
Product Overview
The U2 PSA (Posterior Stabilized Augmentable) Revision Knee system can be used with various augments and extension stems to restore joint stability and address bone loss, allowing surgeons the possibility for an optimal solution for each individual patient.
Product Features




For intact ligament function and minor bone defects.

- Offset adaptor : 2, 4, 6 mm offset
- Straight stem length: 30, 75, 100, 150, 200 mm
- Curved stem length: 150, 200 mm

- Distal & posterior augments: 4, 8 mm thickness
- Distal augment only: 12, 16 mm thickness

- Augment thickness: 5, 10, 15 mm

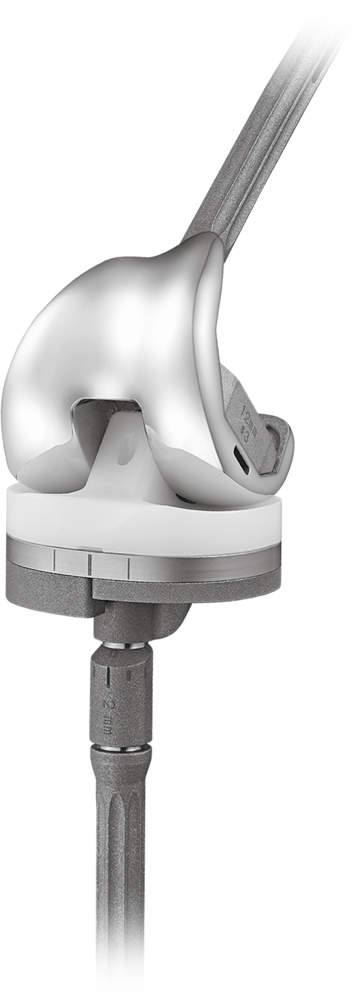
Improved design for optimal function
Enhanced Post-Cam Engagement
Early post and cam engagement (40° flexion) for improved joint stability.
Jump height distance is 21.4 mm to reduce potential for dislocation.
Controlled Femoral Articulation
The femoral component is designed to allow up ± 3.7° of internal/external rotation.
Varus/valgus lift-off is limited to ± 1.4° for increased stability.
Reinforced Femoral Locking Screw
The C-Ring design on the femoral locking screw is intended to prevent loosening/backing out.

Proprietary Tibial Locking Screw
A Built-in reinforcement metal bushing and locking screw within the tibial insert extends into the baseplate. Designed to provide additional structural support and to against shearing force from the femur.

