


Product Overview
The Cellbrick™ Knee Spacer is intended for use in two-stage revision total knee arthroplasty for the management of periprosthetic joint infection (PJI). Its articulating design is intended to maintain joint space and limb length following implant removal, and to support joint mobility during the interim period.
The fenestrated structure is designed to accommodate antibiotic-loaded bone cement, allowing for local antibiotic delivery. An optional intramedullary rod may be used to extend application into the femoral or tibial canal.

Biomechanical & Biological Safety
The UHMWPE femoral and tibial spacer components are designed to provide articulation and structural support during implantation, while helping to maintain soft tissue tension and joint function.

Product Features
Femoral Spacer


Tibial Spacer


Canal Rod



Femoral Spacer Mold


Tibial Spacer Mold
Demonstrated Infection Management
Sustained local gentamicin release maintained above clinically effective levels for 30 days with minimal systemic exposure, supporting infection management in both bench and in-vivo evaluations.
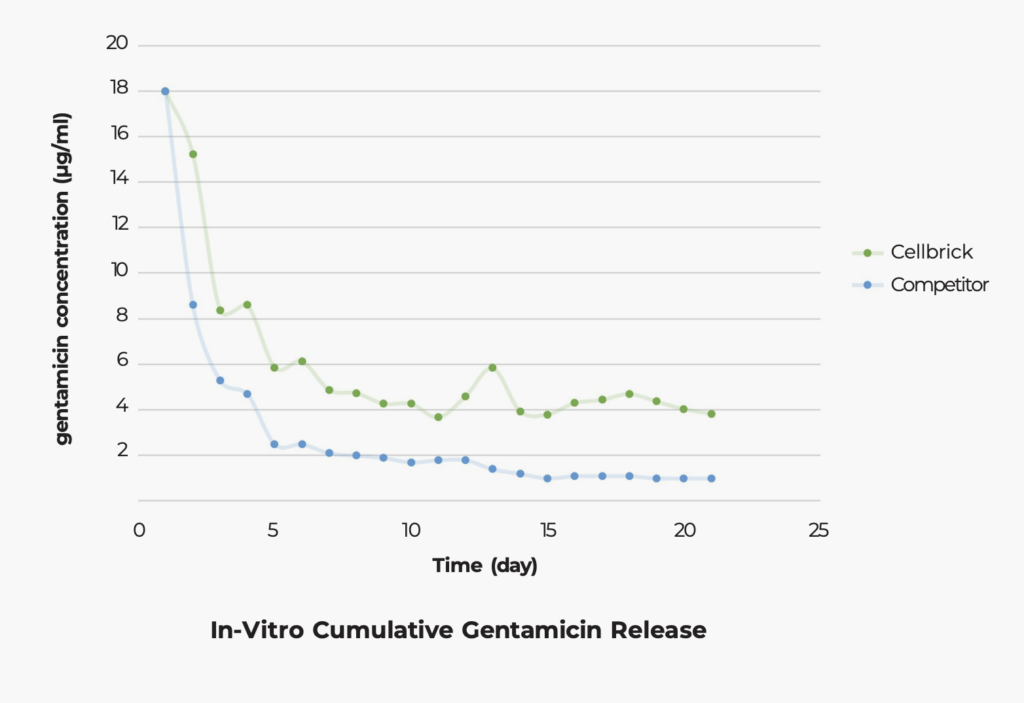
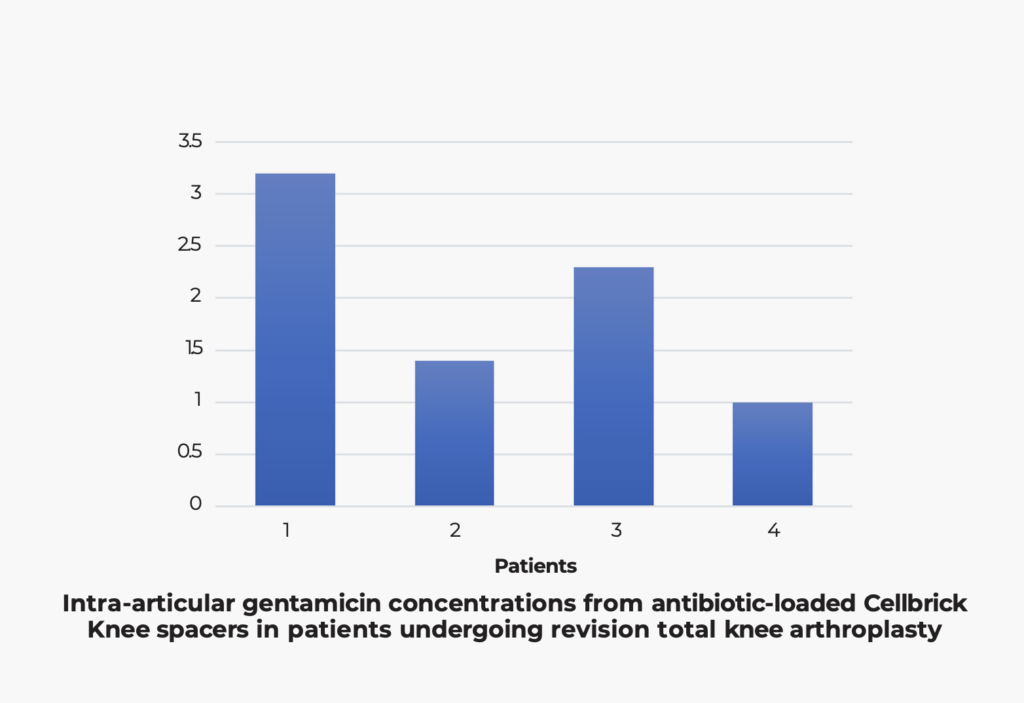
Chang, Y., Lee, M. S., Liau, J. J., Liu, Y. L., Chen, W. C., & Ueng, S. W. (2020). Polyethylene-based knee spacer for infection control: Design concept and pre-clinical in vitro validations. Polymers, 12(10), 2334.
UOC-RD-TR-22050. Data held on file. United Orthopedic Corporation.
UOC-RD-TR-17022. Data held on file. United Orthopedic Corporation.
UOC-UPD-SI-25001. Data held on file. United Orthopedic Corporation.
