


Product Overview
The U2 Knee System’s MDT (Modular Disposable Trial) enables surgeons to complete all trial evaluation surgical technique steps with pre-sterilized, single-use components. This allows for reduced potential of compromised instruments, reduced instrument cleaning/sterilization costs, and faster room turnover.
MDT includes femoral trial sets, tibial trial sets, and patella trial sets designated by implant size

Femoral Trial Set
Includes the Femoral trial, CR notch trial, & PS box trial, as well as the PS notch cutting guide.

Tibial Trial Set
Includes the baseplate trial, PS post trial, and trial spacers to represent tibial construct thicknesses between 9 to 18 mm.

Patellar Trial Set
Includes both onset and inset trial components.
Product Features


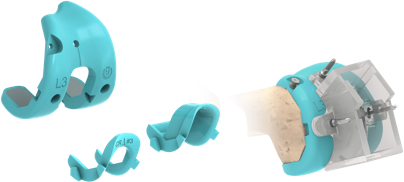

Optimal Surgical Efficiency
When using the U2 Knee System’s AiO Block and MDT Implant Trials together, the number of required instrument trays can be reduced from 6 to 1.5.

